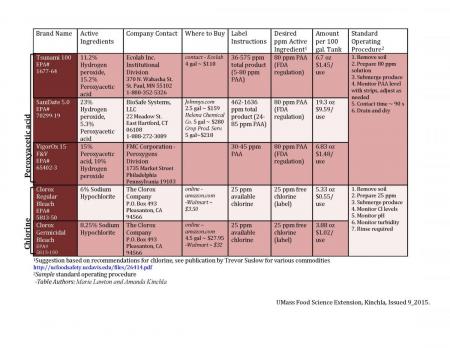
Produce Wash Water Sanitizers: Chlorine and PAA
Food borne pathogens such as Salmonella and Escherichia coli O157:H7 may be present in raw or incompletely composted manure, and when manure is used as a crop amendment, runs off into fields or water, or is carried into fields or processing areas by workers or animals, these pathogens can be passed along to fresh produce. Using a sanitizer in produce wash water can help to maintain clean wash water free of microbial loads. However, choosing and sourcing a sanitizer can be difficult, especially on the small farm scale. This article provides an overview of two effective sanitizing agents – chlorine-based sanitizers and peroxyacetic acid – to help alleviate the stress of deciding which sanitizer is best for certain applications. Note that other types of sanitizers are available such as ozone and chlorine dioxide but the following have been chosen because they are the most readily available for small-scale Northeast growers.
Chlorine-based sanitizers
Chlorine is popularly used as a produce wash water sanitizer due to its documented efficacy and low cost. Though many different chlorine bleach products are available in stores, any product used for this agricultural purpose must be registered with the EPA and specifically labeled for fruit & vegetable washing (1). In the table below, two examples of Clorox bleach are given which are allowed for this use. Certain conditions need to be taken into account when using chlorine, such as the temperature and pH of the wash water as well as the presence of organic matter, since chlorine reacts with organic matter and becomes inactive. Bleach is inexpensive, but managing these factors can lead to added cost. Be aware that the Clorox label states that for fruit and vegetable washing, the concentration of available chlorine in the water may not be more than 25 ppm, and use rates are given accordingly. Fact sheets and other food safety resources may recommend preparing wash solutions at higher concentrations using household bleach, but be sure to follow the label for whichever product you are using. Using free chlorine test strips to measure ppms will help you determine product use rates to stay within the allowable limit. The FDA permits the use of chlorine for food contact, and the EPA regulates it as a general use pesticide.
Peroxyacetic acid
Peroxyacetic acid (PAA) is a combination of acetic acid and hydrogen peroxide. It is effective in reducing the microbial load of wash water by a variety of mechanisms (2). While on the more expensive side for initial cost, peroxyacetic acid does not react with organic matter in the same way chlorine does, and is active over a broad pH range, so dunking solutions don’t have to be changed as frequently between batches (1). The FDA does not permit PAA to exceed 80 ppm in produce wash water, and the EPA regulates PAA as a general use pesticide.
Considerations
This guide is intended to help you make a decision as to which type of sanitizer will work best for you. The total cost per use must take into account both the initial cost of each product, as well as the cost of frequent solution changes and monitoring for conditions such as pH. As discussed, chlorine is reactive to organic matter and may require more per use than indicated in the chart below. Further studies regarding this reactivity will be carried out in the future to compliment this document. You should have a standard protocol that all workers can follow for whichever method you choose, and keep a log book to document sanitizer use rates and conditions such as pH. An example of this kind of log is provided in the UMass GAP manual as a Word document which can be downloaded and tailored for use on your own farm.
Click on table for a larger view.
Sources:
The Center for Agriculture, Food and the Environment and UMass Extension are equal opportunity providers and employers, United States Department of Agriculture cooperating. Contact your local Extension office for information on disability accommodations. Contact the State Center Director’s Office if you have concerns related to discrimination, 413-545-4800 or see ag.umass.edu/civil-rights-information.